Publications
Fecal Immunochemical Test
National guidelines
Halloran S, Launoy G, Zappa M. European Commission. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition – Faecal Occult Blood Testing. Endoscopy 2012; 44:SE65–SE87. PubMed >
National Institute for Health and Care Excellence. NICE guideline NG12. Suspected cancer: recognition and referral. Published 2015. NICE website >
U.S. Preventive Services Task Force. Screening for Colorectal Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2021;325(19):1965-1977. PubMed >
U.S. Multi-Society Task Force. Colorectal Cancer Screening: Recommendations for Physicians and Patients From the U.S. Multi-Society Task Force on Colorectal Cancer. Gastrointestinal Endoscopy, 2017; Volume 153, Issue 1, 307-323. PubMed >
Canadian Task Force on Preventive Health Care. Recommendations on screening for colorectal cancer in primary care. CMAJ Mar 2016, 188 (5) 340-348. PubMed >
Lin JS, Piper M, Perdue LA, et al. Screening for Colorectal Cancer: A Systematic Review for the US Preventive Services Task Force: Evidence Synthesis No. 135. Rockville, MD: Agency for Healthcare Research and Quality; 2016. AHRQ publication 14 05203-EF-1. PubMed >
Screening program
Steele RJ, McDonald PJ, Digby J, et al. Clinical outcomes using a faecal immunochemical test for haemoglobin as a first-line test in a national programme constrained by colonoscopy capacity. United European Gastroenterol J. 2013;1(3):198-205. PubMed >
Quintero E, Castells A, Bujanda L, et al. Colonoscopy versus fecal immunochemical testing in colorectal-cancer screening. N Engl J Med. 2012;366(8):697-706. PubMed >
Zubero MB, Arana-Arri E, Pijoan JI, et al. Population-based colorectal cancer screening: comparison of two fecal occult blood test. Front Pharmacol. 2014;4:175. PubMed >
Faivre J, Dancourt V, Denis B, et al. Comparison between a guaiac and three immunochemical faecal occult blood tests in screening for colorectal cancer. Eur J Cancer. 2012;48(16):2969-76. PubMed >
Rubeca T, Rapi S, Confortini M, et al. Evaluation of diagnostic accuracy of screening by fecal occult blood testing (FOBT). Comparison of FOB Gold and OC Sensor assays in a consecutive prospective screening series. Int J Biol Markers. 2006;21(3):157-61. PubMed >
Chiang TH, Chuang SL, Chen SL, et al. Difference in performance of fecal immunochemical tests with the same hemoglobin cutoff concentration in a nationwide colorectal cancer screening program. Gastroenterology. 2014;147(6):1317-26. PubMed >
van Rossum LG, van Rijn AF, Laheij RJ, et al. Cutoff value determines the performance of a semi-quantitative immunochemical faecal occult blood test in a colorectal cancer screening programme. Br J Cancer. 2009;101(8):1274-81. PubMed >
van Roon AH, Goede SL, van Ballegooijen M, et al. Random comparison of repeated faecal immunochemical testing at different intervals for population-based colorectal cancer screening. Gut. 2013;62(3):409-15. PubMed >
Sali L, Grazzini G, Carozzi F, et al. Screening for colorectal cancer with FOBT, virtual colonoscopy and optical colonoscopy: study protocol for a randomized controlled trial in the Florence district (SAVE study). Trials. 2013 Mar 15;14:74. PubMed >
Santare D, Kojalo I, Huttunen T, et al. Improving uptake of screening for colorectal cancer: a study on invitation strategies and different test kit use. Eur J Gastroenterol Hepatol. 2015;27(5):536–543. PubMed >
Chiu HM, Chen SL, Yen AM, et al. Effectiveness of fecal immunochemical testing in reducing colorectal cancer mortality from the One Million Taiwanese Screening Program. Cancer. 2015;121(18):3221-9. PubMed >
OC-SENSOR performance evaluation
Vilkin A, Rozen P, Levi Z, et al. Performance Characteristics and Evaluation of an Automated-Developed and Quantitative, Immunochemical, Fecal Occult Blood Screening Test. Am J Gastroenterol. 2005;100(11):2519-25. PubMed >
Miki I, Maki H, Ikuhiro M, Yoh H. Evaluation of the analytical performance of the new compact, tabletop, discrete-type automated clinical chemistry analyzer “OC-SENSOR Ceres” for fecal occult blood testing. The Journal of Clinical Laboratory Instruments and Reagents. 2021; 44(3):258-264. “We evaluated the analytical performance of OC-SENSOR Ceres in fecal occult blood quantification and obtained overall good results. “
Hemoglobin concentration and risk of colorectal cancer
Chen LS, Yen AM, Chiu SY, et al. Baseline faecal occult blood concentration as a predictor of incident colorectal neoplasia: longitudinal follow-up of a Taiwanese population-based colorectal cancer screening cohort. Lancet Oncol. 2011;12(6):551-8. PubMed >
Peng SM, Chiu HM, Jen HH, et al. Quantile-based fecal hemoglobin concentration for assessing colorectal neoplasms with 1,263,717 Taiwanese screenees. BMC Med Inform Decis Mak. 2019;19(1):94. PubMed >
Grobbee EJ, Schreuders EH, Hansen BE, et al. Association Between Concentrations of Hemoglobin Determined by Fecal Immunochemical Tests and Long-term Development of Advanced Colorectal Neoplasia. Gastroenterology. 2017;153(5):1251-1259.e2. PubMed >
FIT test number
Goede SL, van Roon AH, Reijerink JC, et al. Cost-effectiveness of one versus two sample faecal immunochemical testing for colorectal cancer screening. Gut. 2013;62(5):727-34. PubMed >
Oort FA, van Turenhout ST, Coupé VM, et al. Double sampling of a faecal immunochemical test is not superior to single sampling for detection of colorectal neoplasia: a colonoscopy controlled prospective cohort study. BMC Cancer. 2011;11:434. PubMed >
FIT and interval cancer
Portillo I, Arana-Arri E, Idigoras I, et al. Colorectal and interval cancers of the Colorectal Cancer Screening Program in the Basque Country (Spain). World J Gastroenterol. 2017;23(15):2731-2742. PubMed >
Digby J, Fraser CG, Carey FA, et al. Interval cancers using a quantitative faecal immunochemical test (FIT) for haemoglobin when colonoscopy capacity is limited. J Med Screen. 2016;23(3):130-4. PubMed >
Comparison with gFOBT
Wilschut JA, Habbema JD, van Leerdam ME, et al. Fecal occult blood testing when colonoscopy capacity is limited. J Natl Cancer Inst. 2011;103(23):1741-51. PubMed >
van Rossum LG, van Rijn AF, Laheij RJ, et al. Random Comparison of Guaiac and Immunochemical Fecal Occult Blood Tests for Colorectal Cancer in a Screening Population. Gastroenterology, 2008;135(1):82-90. PubMed >
Rabeneck L, Rumble RB, Thompson F, et al. Fecal immunochemical tests compared with guaiac fecal occult blood tests for population-based colorectal cancer screening. Can J Gastroenterol. 2012;26(3):131-47. PubMed >
van Rossum LG, van Rijn AF, Verbeek AL, et al. Colorectal cancer screening comparing no screening, immunochemical and guaiac fecal occult blood tests: a cost-effectiveness analysis. Int J Cancer. 2011;128(8):1908-17. PubMed >
Product Series
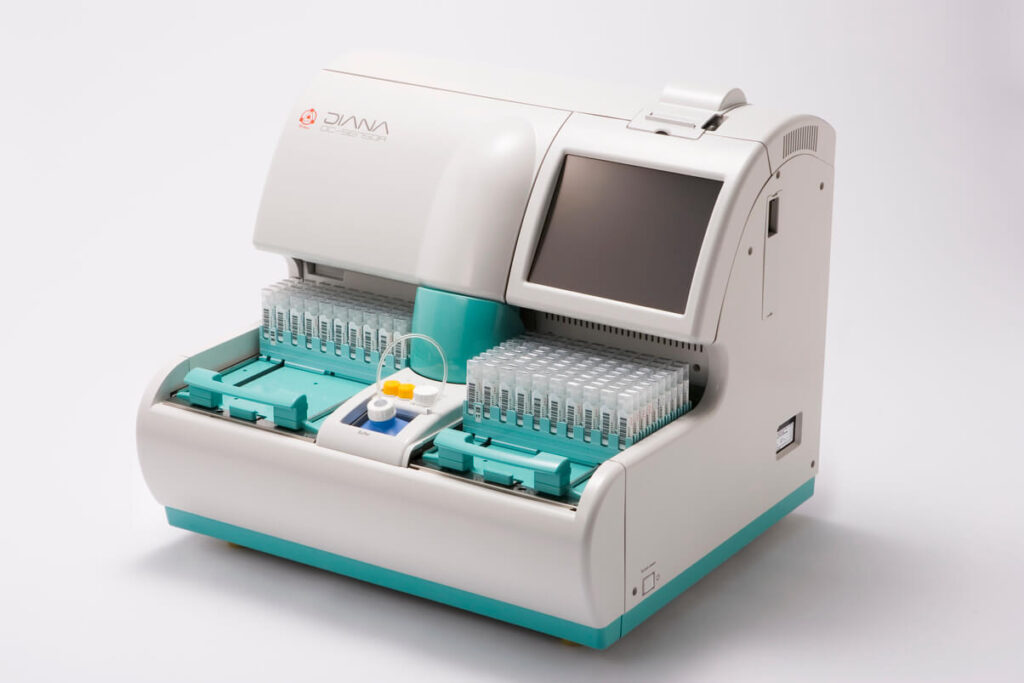
OC-SENSOR® DIANA
OC-SENSOR DIANA is the high-end fecal test analyzer for Fecal immunochemical test. Maximum 250 samples can be performed at one time and the sampling throughput is 280 tests per an hour. The operation is simple as just pressing “start” is required for analyzing.
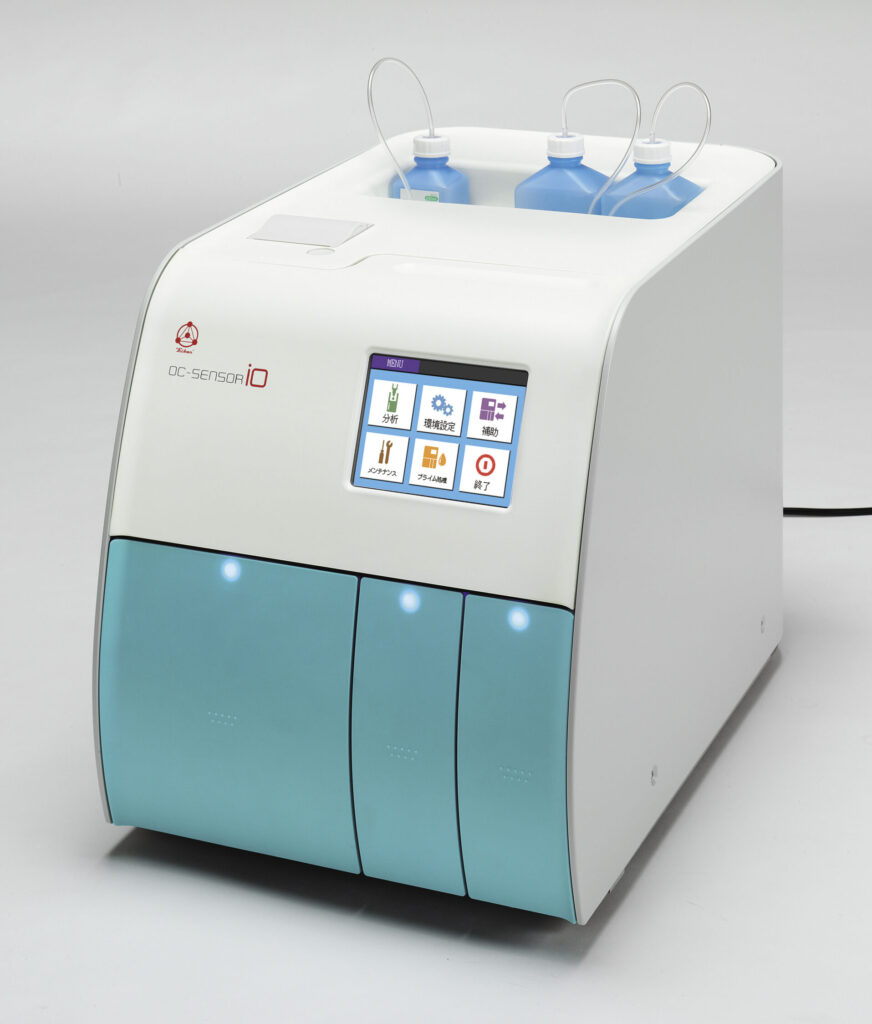
OC-SENSOR® io
OC-SENSOR io is the faecal test analyser with simple to use and compact design for faecal immunochemical test and faecal calprotectin test. Maximum 20 samples can be performed at one time and the sampling throughput is 88 tests per an hour.
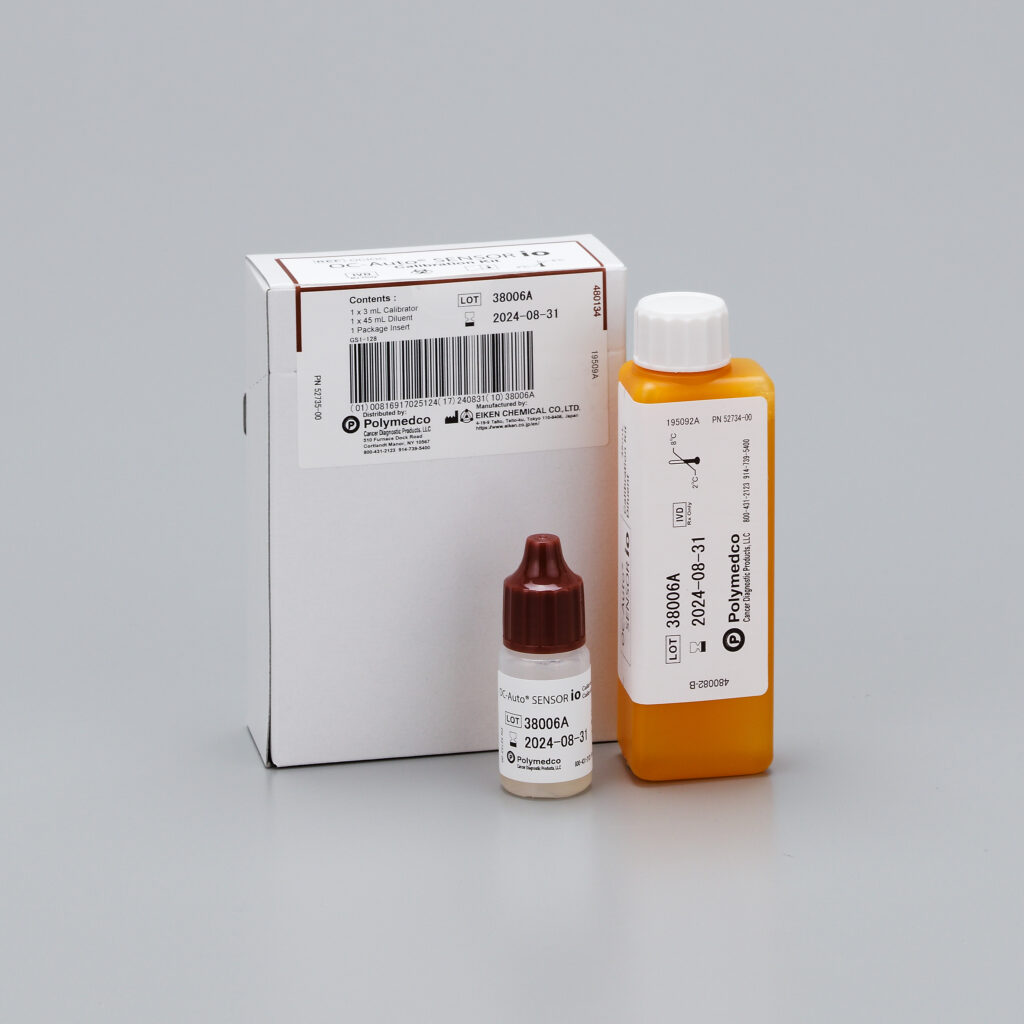
OC-Auto® SENSOR io Calibration Kit
OC-Auto® SENSOR io Calibration Kit is hemoglobin calibration material (hemoglobin concentration of 1000 ng/mL) when using automated immunochemical analyzer, OC-Auto SENSOR io to measure fecal hemoglobin.
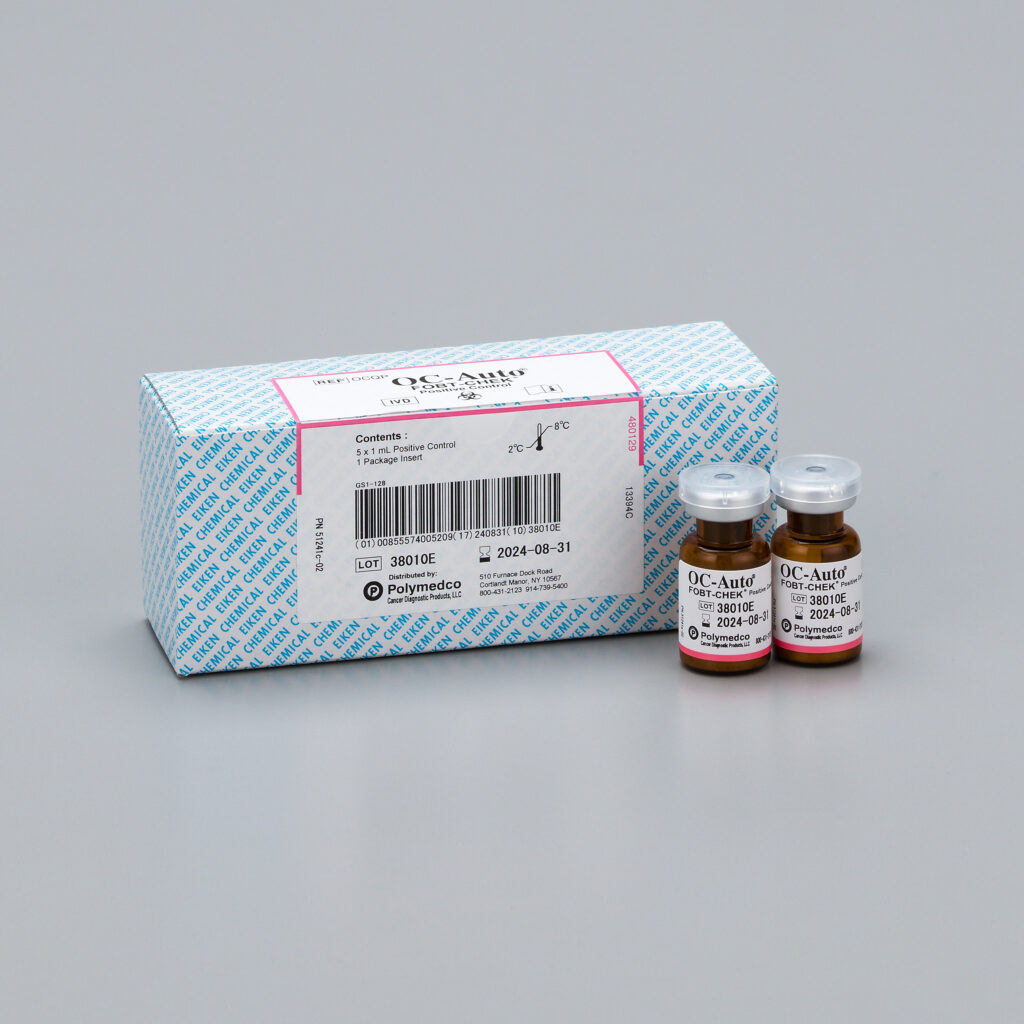
OC-Auto® FOBT CHEK® Positive Control
OC-Auto® FOBT CHEK® Control is a hemoglobin control material designed to monitor FOBT (Fecal Occult Blood Test) procedures. The use of controls helps to monitor technique as well as environmental and physical factors.
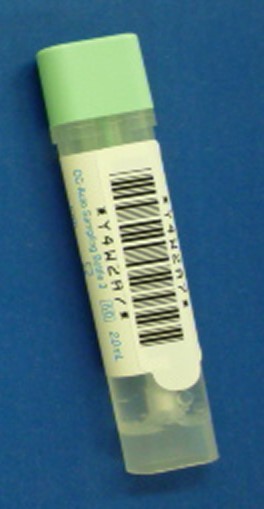
OC-Auto® Sampling Bottle
Ease, Accuracy and Hygiene – All in one compact body. OC-Auto Sampling Bottle has unique characteristics, which provides accurate stool collection. Approximately 100 million tests are performed per year through over 45 countries.
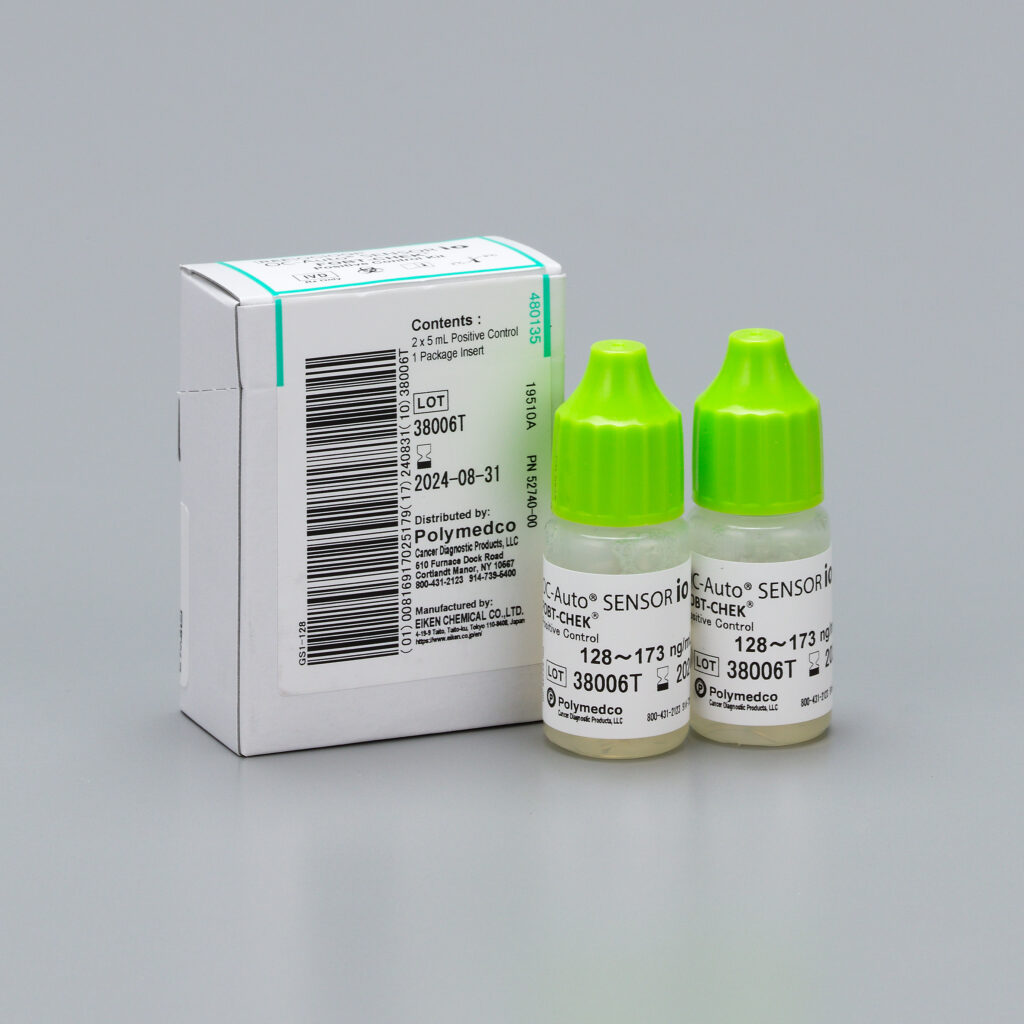
OC-Auto® SENSOR io FOBT CHEK® Positive Control Kit
OC-Auto® SENSOR io FOBT CHEK® Positive Control Kit is a hemoglobin control material designed to monitor iFOBT (Fecal Occult Blood Test) procedures. The use of controls helps to monitor technique as well as environmental and physical factors.
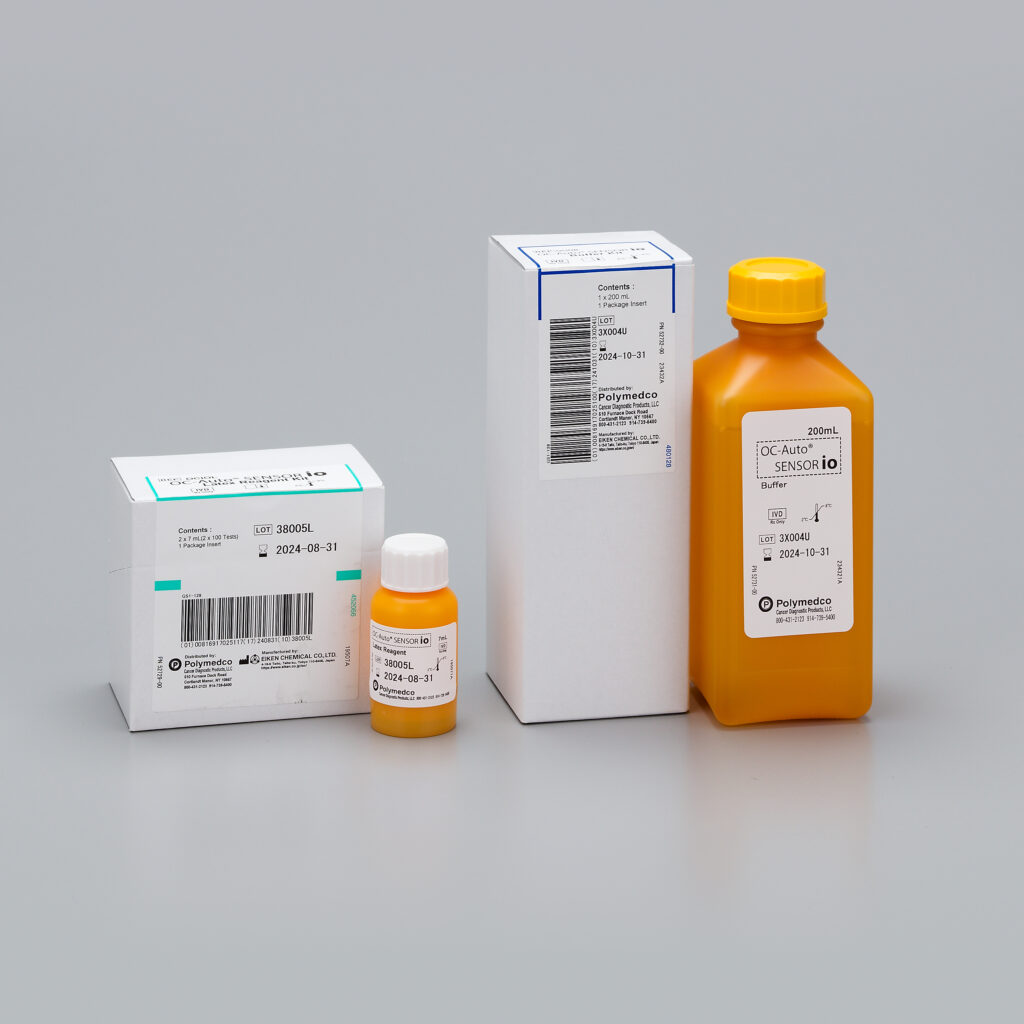
OC-Auto® SENSOR io iFOB Test
OC-Auto® SENSOR io iFOB test is designed to be used together as an immunoassay test system. The test system is intended for the qualitative detection of fecal occult blood in feces by professional laboratories. The automated test is used for measurement of fecal occult blood and is useful an an aid to detect blood in stool when lower gastrointestinal bleeding may be suspected.