Products
Faecal Testing “OC-SENSOR”
30+ years, 45+ countries
OC-SENSOR is the leading Faecal Immunochemical Test. With an extensive body of evidence supporting the platform, OC-SENSOR is recognized worldwide as the standard in colorectal cancer screening and symptomatic triage pathways. When launched in 1989, OC-SENSOR became the first automated faecal occult blood test. Through a continuous platform of innovation, OC-SENSOR has built a reputation of quality and reliability, which in-turn has facilitated the development of regional and national colorectal cancer screening and primary care in over 45 countries.
Product Information
Fully automated fecal test analyzer
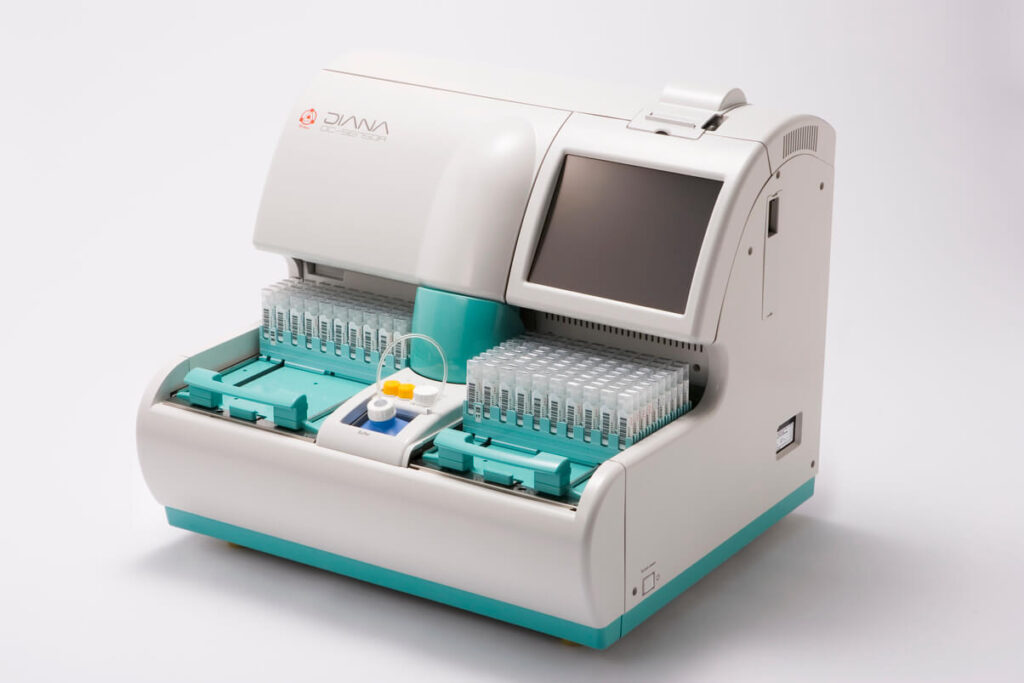
OC-SENSOR® DIANA
OC-SENSOR DIANA is the high-end fecal test analyzer for Fecal immunochemical test. Maximum 250 samples can be performed at one time and the sampling throughput is 280 tests per an hour. The operation is simple as just pressing “start” is required for analyzing.

Fecal Immunochemical Test
Fully automated fecal test analyzer
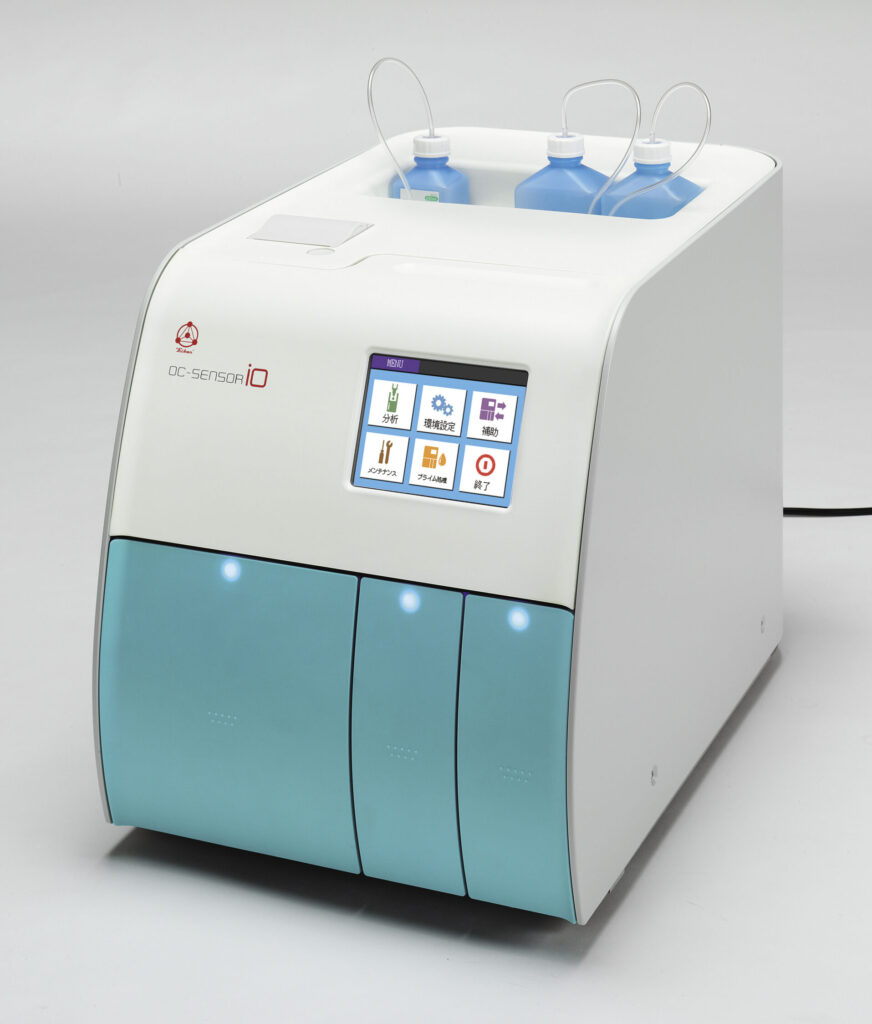
OC-SENSOR® io
OC-SENSOR io is the faecal test analyser with simple to use and compact design for faecal immunochemical test and faecal calprotectin test. Maximum 20 samples can be performed at one time and the sampling throughput is 88 tests per an hour.
Featured Publications of Fecal Immunochemical Test
National guidelines
U.S. Preventive Services Task Force. Screening for Colorectal Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2021;325(19):1965-1977. PubMed >
Halloran S, Launoy G, Zappa M. European Commission. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition – Faecal Occult Blood Testing. Endoscopy 2012; 44:SE65–SE87. PubMed > Publicaions Office of EU >
OC-SENSOR study
Young GP, Symonds EL, Allison JE, et al. Advances in Fecal Occult Blood Tests: The FIT Revolution. Dig Dis Sci (2015) 60:609–622. PubMed >
Allison JE, Fraser CG, Halloran SP, Young GP. Population screening for colorectal cancer means getting FIT: the past, present, and future of colorectal cancer screening using the fecal immunochemical test for hemoglobin (FIT). Gut Liver. 2014;8(2):117–130. PubMed > Publicaions Office of EU >
Product Series
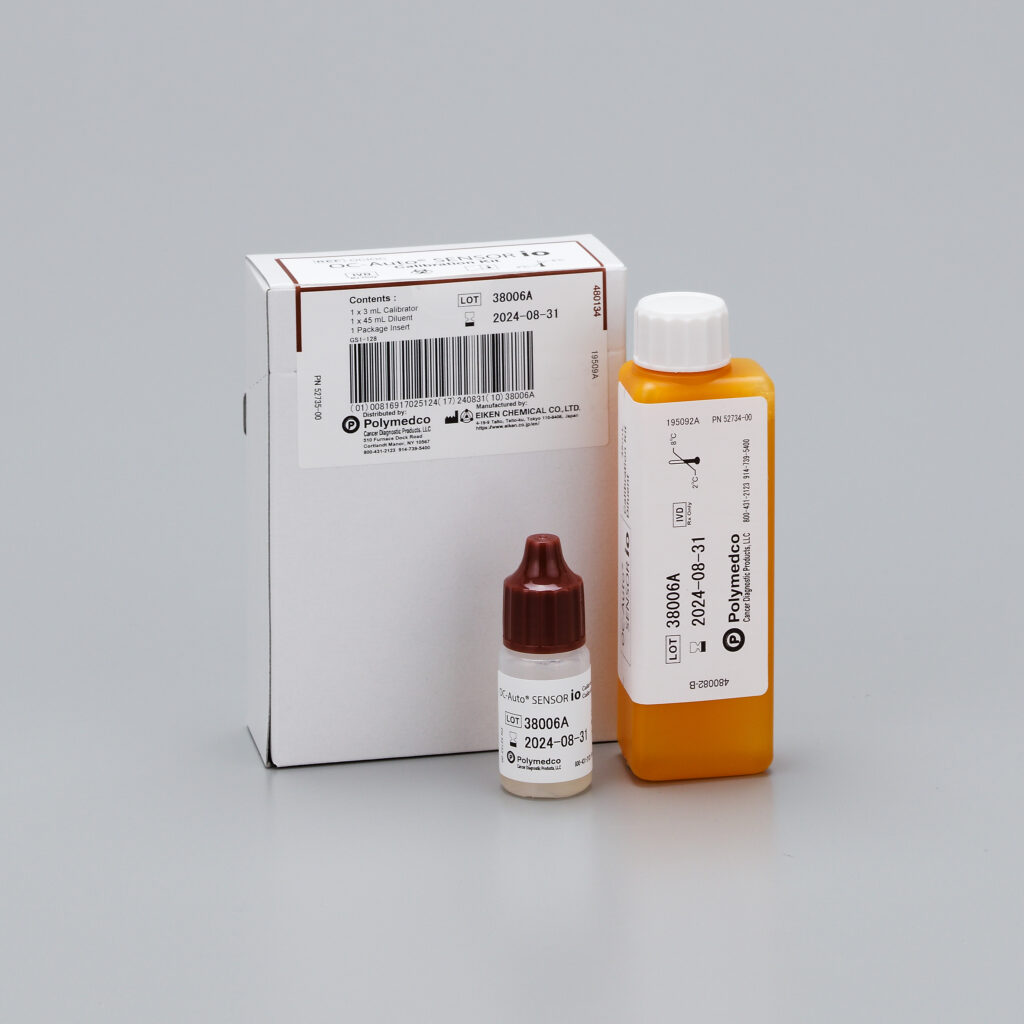
OC-Auto® SENSOR io Calibration Kit
OC-Auto® SENSOR io Calibration Kit is hemoglobin calibration material (hemoglobin concentration of 1000 ng/mL) when using automated immunochemical analyzer, OC-Auto SENSOR io to measure fecal hemoglobin.
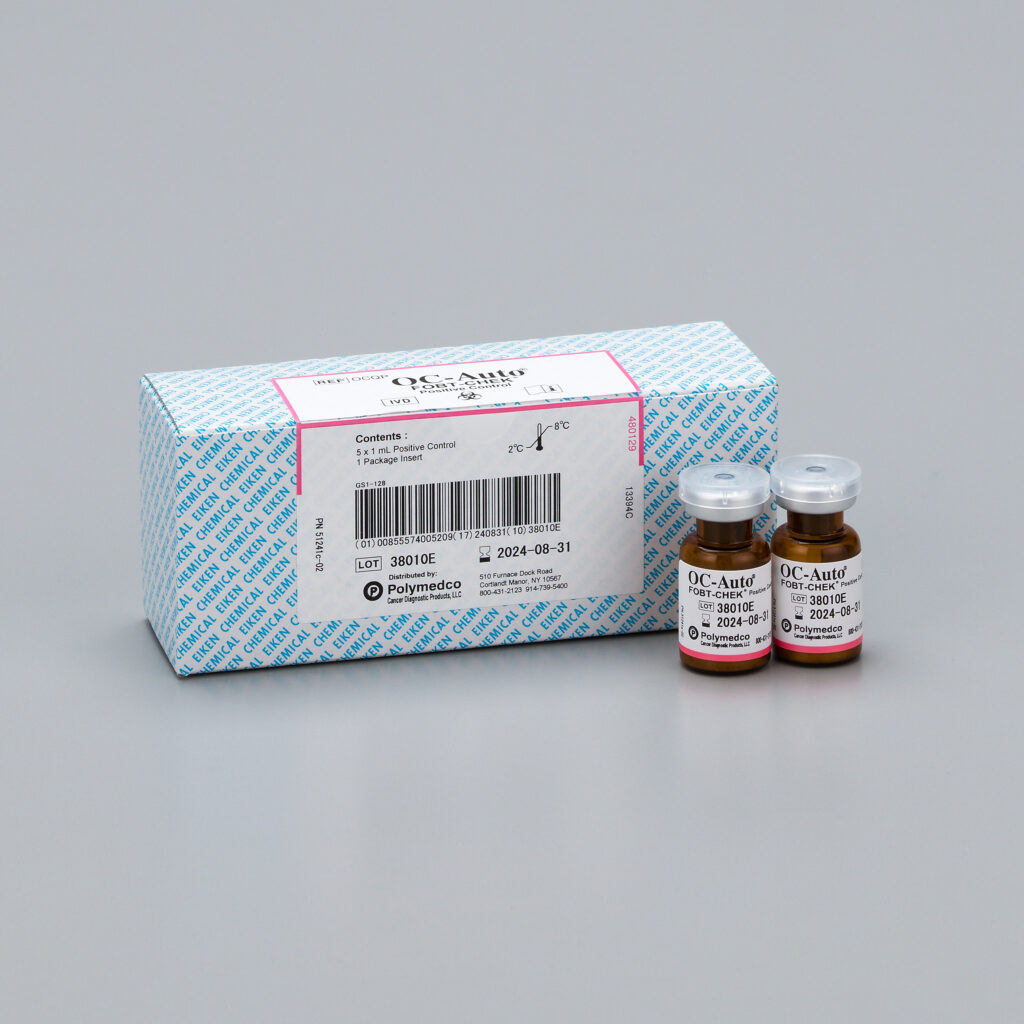
OC-Auto® FOBT CHEK® Positive Control
OC-Auto® FOBT CHEK® Control is a hemoglobin control material designed to monitor FOBT (Fecal Occult Blood Test) procedures. The use of controls helps to monitor technique as well as environmental and physical factors.
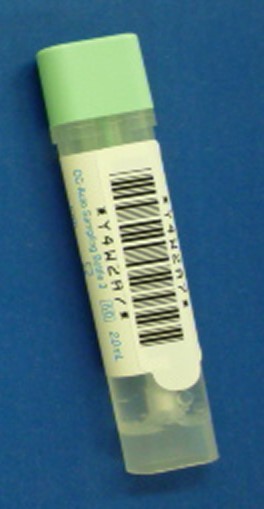
OC-Auto® Sampling Bottle
Ease, Accuracy and Hygiene – All in one compact body. OC-Auto Sampling Bottle has unique characteristics, which provides accurate stool collection. Approximately 100 million tests are performed per year through over 45 countries.
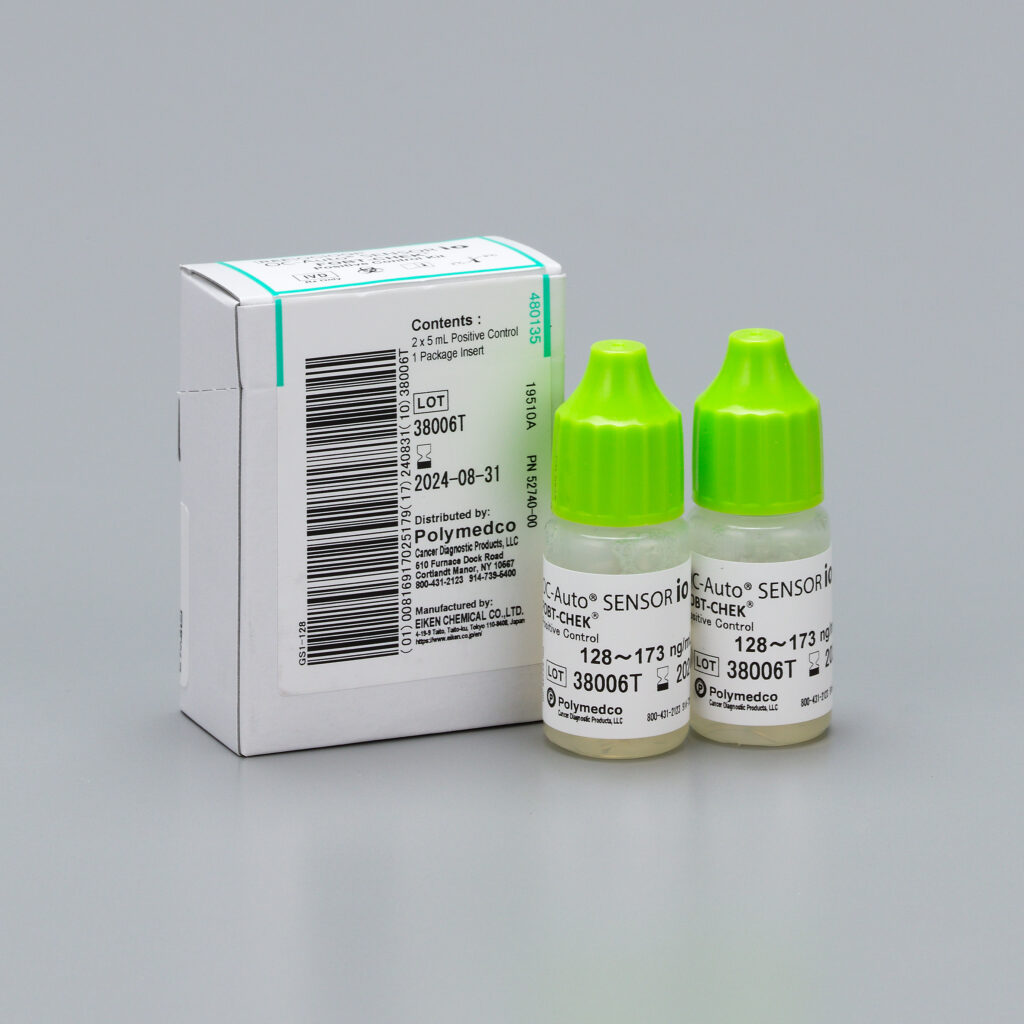
OC-Auto® SENSOR io FOBT CHEK® Positive Control Kit
OC-Auto® SENSOR io FOBT CHEK® Positive Control Kit is a hemoglobin control material designed to monitor iFOBT (Fecal Occult Blood Test) procedures. The use of controls helps to monitor technique as well as environmental and physical factors.
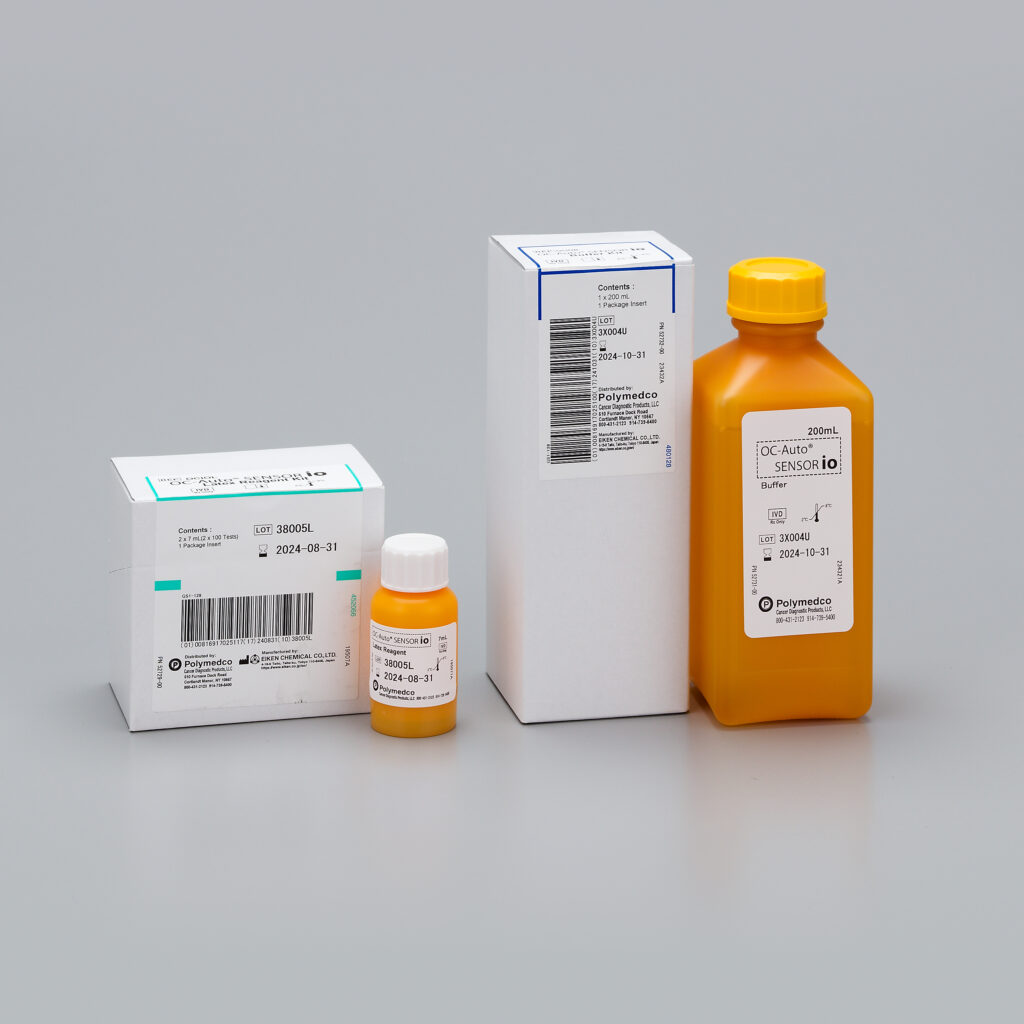
OC-Auto® SENSOR io iFOB Test
OC-Auto® SENSOR io iFOB test is designed to be used together as an immunoassay test system. The test system is intended for the qualitative detection of fecal occult blood in feces by professional laboratories. The automated test is used for measurement of fecal occult blood and is useful an an aid to detect blood in stool when lower gastrointestinal bleeding may be suspected.
CONTACT US
Thank you for your interest in EIKEN CHEMICAL and our products.
Please provide your details below, and we will respond promptly to your inquiry.